What is the second law of thermodynamics

For example, when a path for conduction and radiation is made available, heat always flows spontaneously from a hotter to a colder body. It asserts that a natural process runs only in one sense, and is not reversible. The second law is concerned with the direction of natural processes. The first law of thermodynamics provides the definition of the internal energy of a thermodynamic system, and expresses the law of conservation of energy. Heat flowing from hot water to cold water
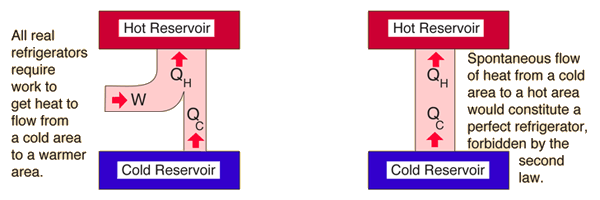
The second law may be formulated by the observation that the entropy of isolated systems left to spontaneous evolution cannot decrease, as they always arrive at a state of thermodynamic equilibrium, where the entropy is highest at the given internal energy.

It can be used to predict whether processes are forbidden despite obeying the requirement of conservation of energy as expressed in the first law of thermodynamics and provides necessary criteria for spontaneous processes. The second law of thermodynamics establishes the concept of entropy as a physical property of a thermodynamic system.
